Access the tools you need, wherever you are.
oVi, now shipping worldwide.
Anaesthetics
Messenger RNA
GLP-1
Radiotherapy
Biologics
CAR T Cell Therapy
Biodefense
Diagnostics
Generics
Ophthalmic
CRISPR
BioTechnology
Pharmaceuticals
Vaccines
Stem Cell
Hyaluronic Acid
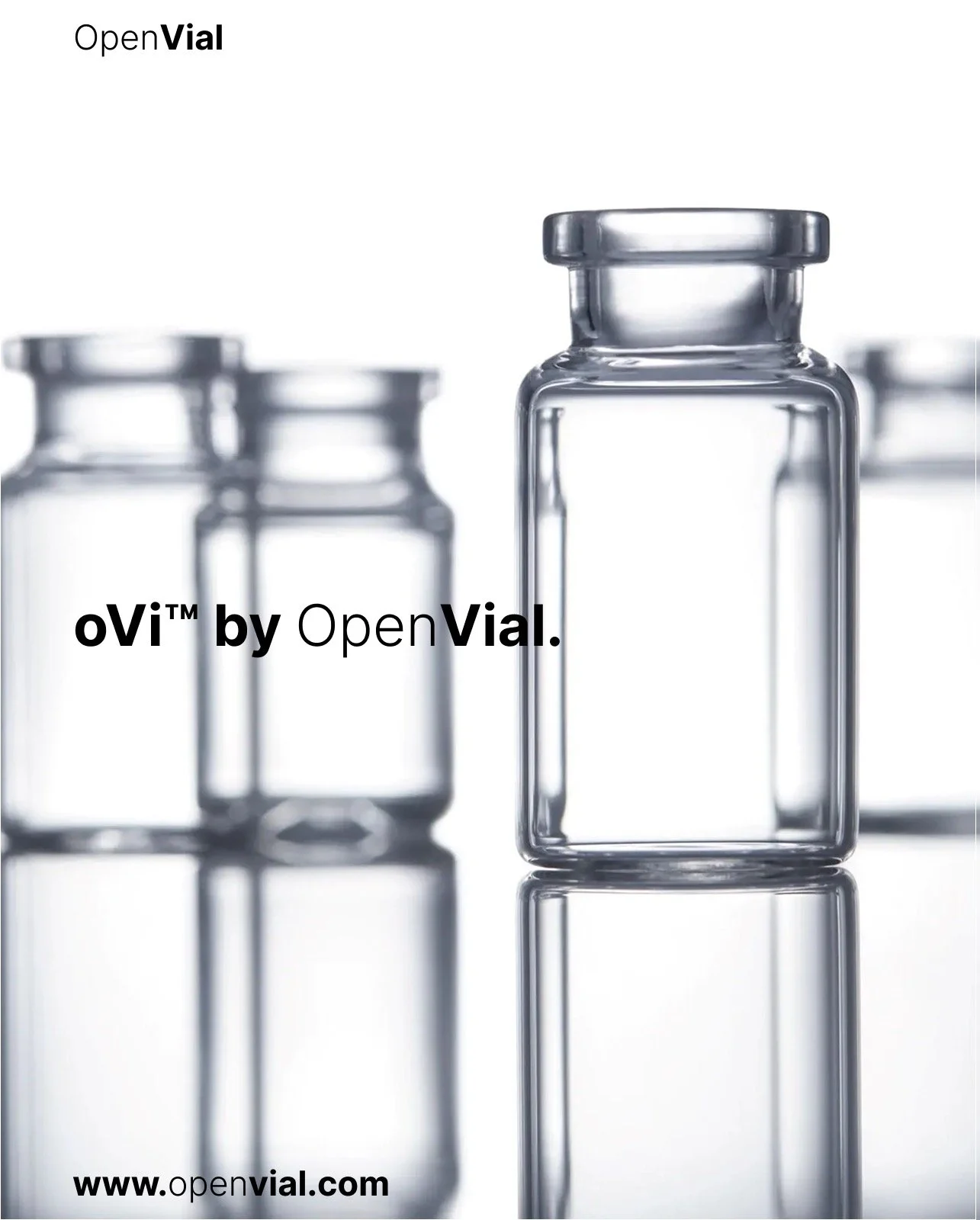
oVI by OpenVial.
oVI by OpenVial is a sterile, pre-tested, double-packed vial system engineered to support early-stage research and development in healthcare.
Sterile : Manufactured in ISO 5 cleanrooms and sterilized per ISO 11137, ensuring a contamination-free environment suitable for aseptic filling and handling.
Pyrogen-Free : Rigorously tested to meet USP <85> and EP 2.6.14 standards, eliminating risks of pyrogen-induced immune responses, making it ideal for injectable solutions and sensitive biologicals.
Particle-Free : Adheres to ISO 14644 standards, with advanced inspection systems ensuring freedom from visible and sub-visible particles, critical for injectable and ophthalmic formulations.
Non-Cytotoxic : Certified non-cytotoxic per ISO 10993-5, ensuring compatibility with biologic formulations and supporting cellular research and therapeutic development.
Double-Packaged Design: Complies with ISO 11607, featuring a proprietary double-packaging system that protects against environmental contaminants and supports aseptic operations.
Global Distribution: Available for worldwide shipment, adhering to regional regulatory standards, with a robust logistics network ensuring secure and timely delivery.
Type 1 Glass : Made from Type 1 R Borosilicate Glass, known for superior chemical and thermal resistance, meeting USP, EP, and JP standards, and available in multiple sizes.
21 Units per Box: Each box contains 21 sterile vials, individually packed to support non-simultaneous use, minimizing handling risks and offering flexibility for varying project timelines.
EO Residual-Free: Ensures purity and safety by validating that vials are free of ethylene oxide residues, suitable for critical pharmaceutical and biotech applications.
OVI by OpenVial delivers a seamless solution tailored to the needs of pioneering research, streamlining workflows and ensuring uncompromising reliability for companies pushing the boundaries of healthcare.
OpenVial creates tools to simplify healthcare innovation.
Traditional pharmaceutical vials manufacturers have optimised their products and operations around industrial aseptic filling. As a consequence, early stage pharmaceutical research center had to adapt their day-to-day operations. And that’s costing them a lot.
OpenVial understands that early stage research in Pharmaceuticals & Biotechnology requires to un-scheduled needs and wants. And that timing requirements for available ressources are volatile.
That’s why OpenVial offers pharmaceutical vials designed specifically for companies tackling complex research.